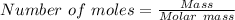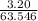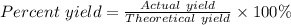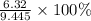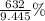Chemistry, 02.12.2020 01:00 angelica7773
Suppose 3.20 g of copper are reacted with excess nitric acid according to the given equation, and 6.32 g Cu(NO3)2 product are obtained.
Cu(s) + 4 HNO3 (aq) --> Cu(NO3)2 (aq) + 2 NO2 (g) + 2 H2O(l)
What is the theoretical yield of Cu(NO3)2? In g
What is the percent yield of Cu(NO3)2? In %

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1


Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
Suppose 3.20 g of copper are reacted with excess nitric acid according to the given equation, and 6....
Questions


Physics, 13.02.2020 18:38

Business, 13.02.2020 18:38



Biology, 13.02.2020 18:38

Engineering, 13.02.2020 18:38