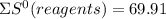Chemistry, 02.12.2020 17:10 BrianKeokot7700
a) Calculate the standard reaction entropy for the combustion of methane to carbon dioxide and liquid water at 298 K. b) Calculate the standard reaction entropy for the hydrolysis of liquid water to form oxygen and hydrogen gas. c) Do your results in (a) and (b) agree with the general rule that reaction entropies are positive if there is a net formation of gas in a reaction, and negative if the is a net reduction of gas?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
List four observations that indicate that a chemical reaction may be taking place
Answers: 1

Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3

Chemistry, 22.06.2019 07:00
The variability in marine salinity between habitats does not impact the fish living there. select the best answer from the choices provided t f
Answers: 1

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3
You know the right answer?
a) Calculate the standard reaction entropy for the combustion of methane to carbon dioxide and liqui...
Questions

Mathematics, 31.03.2021 21:20


English, 31.03.2021 21:20

Computers and Technology, 31.03.2021 21:20



Arts, 31.03.2021 21:20



Social Studies, 31.03.2021 21:20

English, 31.03.2021 21:20

English, 31.03.2021 21:20


Chemistry, 31.03.2021 21:20

Arts, 31.03.2021 21:20

English, 31.03.2021 21:20

Mathematics, 31.03.2021 21:20

Mathematics, 31.03.2021 21:20

English, 31.03.2021 21:20

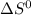 = - 242.2J/K.mol
= - 242.2J/K.mol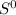 .
. 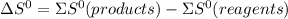
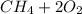 ⇒
⇒ 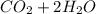
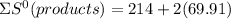 = 353.8
= 353.8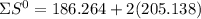 = 596.54
= 596.54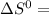 353.8 - 596.54
353.8 - 596.54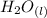 ⇒
⇒ 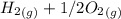
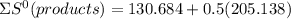 = 233.253
= 233.253