
Chemistry, 03.01.2020 02:31 audreymarie2940
Calculate the hydrogen-ion concentration [h+] for the aqueous solution in which [oh–] is 1 x 10–11 mol/l. is this solution acidic, basic, or neutral? show all work.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 12:30
Clyde and marilyn are riding a roller coaster. during which section(s) of the track is their potential energy converted to kinetic energy? a. from point b to point c only b. from point b to point d only c. from point a to point b only d. from point a to point b and from point c to point d
Answers: 1

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 22.06.2019 18:00
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1
You know the right answer?
Calculate the hydrogen-ion concentration [h+] for the aqueous solution in which [oh–] is 1 x 10–11 m...
Questions




Mathematics, 12.02.2021 05:00


Spanish, 12.02.2021 05:00

Mathematics, 12.02.2021 05:00

Business, 12.02.2021 05:00

Geography, 12.02.2021 05:00



Mathematics, 12.02.2021 05:00



Mathematics, 12.02.2021 05:00

Mathematics, 12.02.2021 05:00



Chemistry, 12.02.2021 05:00

![[H+][OH-]= Kw = 1.0 x 10^-14](/tpl/images/0440/7437/9a04c.png)
![[H+]= Kw/ [OH-]= 1.0x 10^-14](/tpl/images/0440/7437/e7cfa.png)
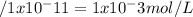
![pH = - log [H+]= - log 1 x 10^-3 = 3 \ \textless \ 7](/tpl/images/0440/7437/22063.png)


