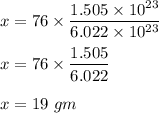Chemistry, 02.12.2020 23:30 giigigihyys2111
What would be the mass, in grams, of 1.505 x 10^23 molecules of carbon disulfide (CS2)?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 22.06.2019 00:00
The p sub shell can hold up to 8 electrons in an atom. true or false?
Answers: 1

Chemistry, 22.06.2019 14:30
Chemistry worksheet - i am not sure what they are asking for exactly?
Answers: 1

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3
You know the right answer?
What would be the mass, in grams, of 1.505 x 10^23 molecules of carbon disulfide (CS2)?...
Questions

Mathematics, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30


Mathematics, 24.11.2020 18:30

English, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30



Mathematics, 24.11.2020 18:30



Mathematics, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30


English, 24.11.2020 18:30


Mathematics, 24.11.2020 18:30


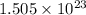 molecules of carbon disulfide (CS₂).
molecules of carbon disulfide (CS₂).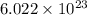 molecules of CS₂ is 76 gm/mol.
molecules of CS₂ is 76 gm/mol.