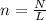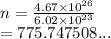Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 23.06.2019 03:30
In chemistry, the type of an atom (what element it is) is determined by: a) the number of protons it contains in its nucleus.b) the number of neutrons it contains in its nucleus.c) the number of protons it has in a cloud around the nucleus.d) the number of neutrons it has in a cloud around the nucleus.e) the number of electrons it exchanges with its neighbors.
Answers: 1

Chemistry, 23.06.2019 04:10
Two solids are mixed in a flask and stirred. after a few minutes, the flask becomes cold. which of the following best describes this reaction? a. an exothermic reaction b. a combustion reaction c. an endothermic reaction d. a decomposition reaction
Answers: 1
You know the right answer?
How many moles are in 4.67 x 10^26 atoms of Iron?...
Questions

History, 10.11.2020 19:40

Mathematics, 10.11.2020 19:40

Mathematics, 10.11.2020 19:40

English, 10.11.2020 19:40



History, 10.11.2020 19:40

Spanish, 10.11.2020 19:40



Mathematics, 10.11.2020 19:40

History, 10.11.2020 19:40




Arts, 10.11.2020 19:40



History, 10.11.2020 19:40

English, 10.11.2020 19:40