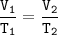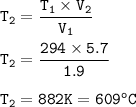Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 05:00
Choose all the answers that apply. ionic compounds dissolve easily in water do not dissolve in water have low melting points have high melting points conduct electricity when melted
Answers: 1


Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
The volume of gas in a balloon is 1.90 L at 21.0o C. The balloon is heated, causing it to expand to...
Questions

History, 25.02.2021 17:30


History, 25.02.2021 17:30



History, 25.02.2021 17:30


Mathematics, 25.02.2021 17:30

Mathematics, 25.02.2021 17:30


Mathematics, 25.02.2021 17:30

Mathematics, 25.02.2021 17:30

Mathematics, 25.02.2021 17:30


Mathematics, 25.02.2021 17:30


Mathematics, 25.02.2021 17:30


Mathematics, 25.02.2021 17:30

Health, 25.02.2021 17:30