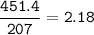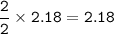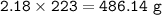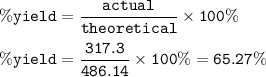Consider the reaction.
2Pb(s)+O2(g)⟶2PbO(s)
An excess of oxygen reacts with 451.4 g of...


Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:10
How is 0.00235 expressed in proper scientific notation? a. 2.35 × 10-3 b. 0.235 × 10-2 c. 2.35 d. 2.35 × 103
Answers: 1

Chemistry, 21.06.2019 22:30
Llama have 74 chromosomes how many chromosomes will they be found in their gametes explain how you know
Answers: 2

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1
You know the right answer?
Questions


History, 05.05.2020 06:43

Mathematics, 05.05.2020 06:43

Chemistry, 05.05.2020 06:43

Social Studies, 05.05.2020 06:43



History, 05.05.2020 06:43


Physics, 05.05.2020 06:43





Physics, 05.05.2020 06:43



Mathematics, 05.05.2020 06:43

Mathematics, 05.05.2020 06:43