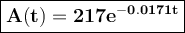Chemistry, 07.12.2020 14:00 lucerogd170
The amount of 217 mg of an isotope is given by A(t) = 217 € -0.0171, where t is time in years since the initial amount of 217 mg was present. Find the amount to the nearest milligraM left after 20 years

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2


Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3
You know the right answer?
The amount of 217 mg of an isotope is given by A(t) = 217 € -0.0171, where t is time in years since...
Questions

Health, 23.07.2019 07:00




Mathematics, 23.07.2019 07:10


Mathematics, 23.07.2019 07:10



English, 23.07.2019 07:10


English, 23.07.2019 07:10



Mathematics, 23.07.2019 07:10



Biology, 23.07.2019 07:10

Medicine, 23.07.2019 07:10