
Chemistry, 09.12.2020 22:20 beeboppity
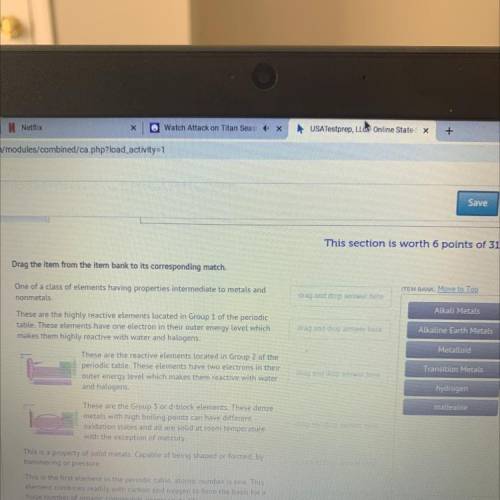
One of a class of elements having properties intermediate to metals and nonmetals.
These are the highly reactive elements located in Group 1 of the periodic table. These elements have one electron in their outer energy level which makes them highly reactive with water and halogens.
These are the reactive elements located in Group 2 of the periodic table. These elements have two electrons in their outer energy level which makes them reactive with water and halogens.
These are the Group 3 or d-block elements. These dense metals with high boiling points can have different oxidation states and all are solid at room temperature with the exception of mercury.
This is a property of solid metals. Capable of being shaped or formed, by hammering or pressure.
This is the first element in the periodic table, atomic number is one. This element combines readily with carbon and oxygen to form the basis for a huge number of organic compounds necessary to life.

Answers: 2
Another question on Chemistry


Chemistry, 21.06.2019 23:50
2points what is the job of a scientist? a. to answer ethical questions. b. to write laws based on his or her knowledge. c. to ask and answer scientific questions. d. to ignore facts that do not support his or her theory.
Answers: 1

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 20:30
What is a difference between a mixture of elements and a mixture of compounds
Answers: 1
You know the right answer?
One of a class of elements having properties intermediate to metals and nonmetals.
These are the hi...
Questions

Mathematics, 22.05.2021 08:30

English, 22.05.2021 08:30



Biology, 22.05.2021 08:30

Biology, 22.05.2021 08:30


Mathematics, 22.05.2021 08:30


Mathematics, 22.05.2021 08:30

Mathematics, 22.05.2021 08:30


Social Studies, 22.05.2021 08:30

History, 22.05.2021 08:30


Social Studies, 22.05.2021 08:30

Mathematics, 22.05.2021 08:30



History, 22.05.2021 08:30



