
Ammonia (A) diffuses through a stagnant layer of air (B), 1cm thick, at 25 ºC and 1 atm total pressure. The partial pressures of ammonia on the two sides of the air layer are: PA0=0.9 atm and PAl=0.1 atm respectively. Air is none diffusing. Calculate the molar flux of ammonia. DAB= 0.214 cm2 /s

Answers: 1
Another question on Chemistry

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 1

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1

Chemistry, 23.06.2019 04:31
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1

Chemistry, 23.06.2019 08:00
The goal of this experiment was to answer the question "what is the effect of a gas' temperature on its volume? " you formulated the hypothesis below. hypothesis: if a fixed amount of gas is heated, then the volume will increase because the heat will cause the molecules of gas to move faster and further apart. to test this hypothesis, you changed the of the gas between 0 and 100°c (273 and 373 k) and calculated the resulting of the gas.
Answers: 2
You know the right answer?
Ammonia (A) diffuses through a stagnant layer of air (B), 1cm thick, at 25 ºC and 1 atm total pressu...
Questions

Mathematics, 27.11.2020 18:20

World Languages, 27.11.2020 18:20

English, 27.11.2020 18:20

Arts, 27.11.2020 18:20


Chemistry, 27.11.2020 18:20

Mathematics, 27.11.2020 18:20





English, 27.11.2020 18:20

Mathematics, 27.11.2020 18:20


Physics, 27.11.2020 18:20

Biology, 27.11.2020 18:30


History, 27.11.2020 18:30

Mathematics, 27.11.2020 18:30

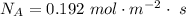
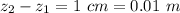
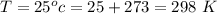
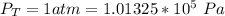
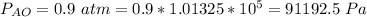
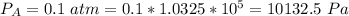
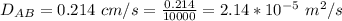
![N_A = \frac{D_{AB} * P_T }{RT(z_2 -z_1)} * ln [\frac{P_T - P_{Al}}{P_T - P_{AO}} ]](/tpl/images/0969/5009/086d8.png)
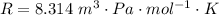
![N_A = \frac{2.14 *10^{-5} * 1.01325*10^{5} }{8.314 *298 (0.01)} * ln [\frac{1 - 0.1}{1 - 0.9} ]](/tpl/images/0969/5009/c7bba.png)


