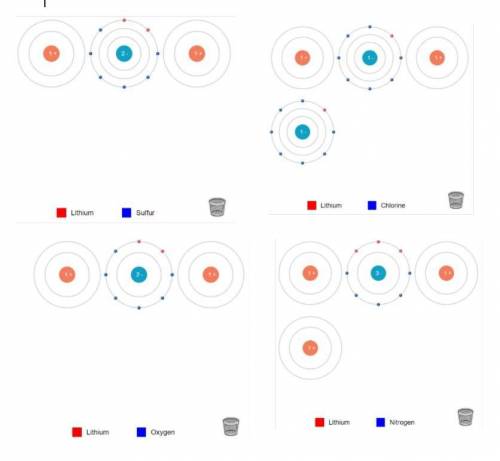
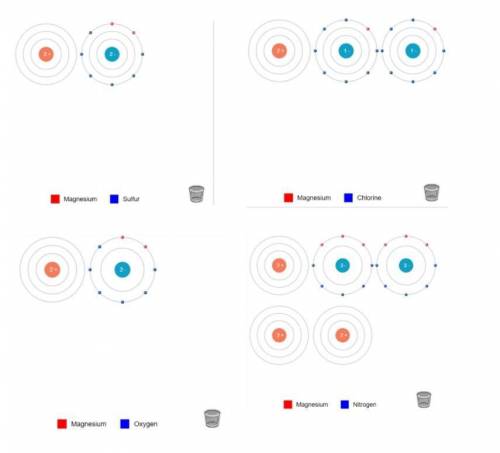
Can someone help me with my chemistry homework please???
...

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:40
Achange in the number of neutrons in an atom will change an blank . when the number of protons changes in an atom, a new element will form.
Answers: 2


Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3
You know the right answer?
Questions


Mathematics, 04.03.2022 17:30









Arts, 04.03.2022 17:40

Mathematics, 04.03.2022 17:40

Mathematics, 04.03.2022 17:40


Mathematics, 04.03.2022 17:40

Spanish, 04.03.2022 17:40


English, 04.03.2022 17:40

Mathematics, 04.03.2022 17:40

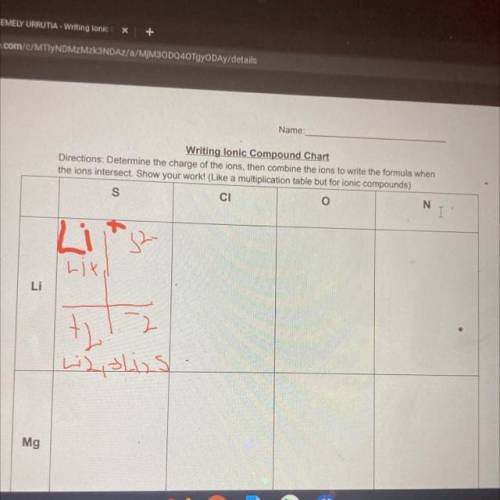
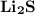 Ion Charges:
Ion Charges: 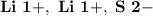
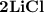 Ion Charges:
Ion Charges: 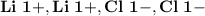
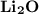 Ion Charges:
Ion Charges: 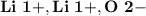
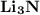 Ion Charges:
Ion Charges: 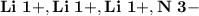
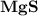 Ion Charges:
Ion Charges: 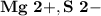
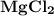 Ion Charges:
Ion Charges: 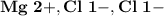
 Ion Charges:
Ion Charges: 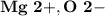
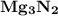 Ion Charges:
Ion Charges: 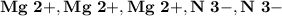 Explanation:
Explanation: