
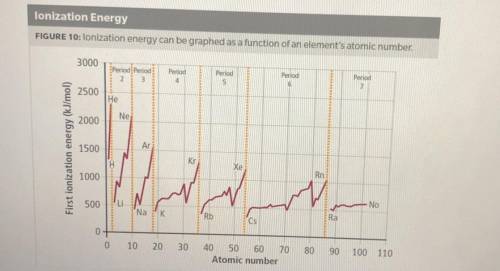
1. How does ionization energy change with atomic number? Use evidence from the graph to support your claim.
2. How does ionization energy change across a period and down a group on the periodic table? Use evidence from the graph to support your claim.
3. What describes an effect on ionization energy when moving down a group? Select all correct answers.
A. The ionization energy increases down a group
B. The ionization energy decreases down a group
C. The valence electrons are in energy levels farther from the nucleus
D. The shielding effect is less

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 04:30
What are the three major branches of natural science? • earth and space science, life science, physical science •earth and space science, physical science, chemistry •physical science, life science, chemistry •life science, chemistry, physics
Answers: 1


Chemistry, 22.06.2019 20:00
How are the terms group and period used on the periodic table
Answers: 1
You know the right answer?
1. How does ionization energy change with atomic number? Use evidence from the graph to support your...
Questions

Mathematics, 11.11.2020 03:40


Social Studies, 11.11.2020 03:40



Mathematics, 11.11.2020 03:40

History, 11.11.2020 03:40

Biology, 11.11.2020 03:40



Mathematics, 11.11.2020 03:40

Mathematics, 11.11.2020 03:40


Mathematics, 11.11.2020 03:40


Spanish, 11.11.2020 03:40

Business, 11.11.2020 03:40

Mathematics, 11.11.2020 03:40

Social Studies, 11.11.2020 03:40

Mathematics, 11.11.2020 03:40



