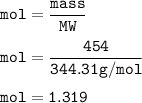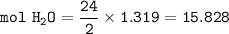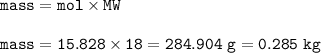Chemistry, 12.12.2020 17:00 miguegen6225
454 grams of isomaltitol, C12H24O11, was combusted in a furnace completely in an excess oxygen. The products of this reaction are water and carbon dioxide. What is the theoretical yield (in kilograms) of water produced from this reaction?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
Initially, the balloon had 3.0 liters of gas at a pressure of 400 kpa and was at a temperature of 294 k. if the balloon is cooled to 277 k and its volume decreased to 1 l, what will the new pressure in the balloon be?
Answers: 1

Chemistry, 22.06.2019 13:00
The number of neutrons is equal to the atomic number minus the atomic mass. a. true b. false
Answers: 2


Chemistry, 23.06.2019 03:30
If you need to add 27.50ml of a solution, which piece of glassware would you use to deliver this volume and explain how you would determine if the 27.50 ml was measured?
Answers: 1
You know the right answer?
454 grams of isomaltitol, C12H24O11, was combusted in a furnace completely in an excess oxygen. The...
Questions

Mathematics, 03.11.2020 01:00

Mathematics, 03.11.2020 01:00

Mathematics, 03.11.2020 01:00

Mathematics, 03.11.2020 01:00




Arts, 03.11.2020 01:00


Mathematics, 03.11.2020 01:00


Mathematics, 03.11.2020 01:00


Mathematics, 03.11.2020 01:00



World Languages, 03.11.2020 01:00

English, 03.11.2020 01:00