
Chemistry, 12.12.2020 17:10 janayshas84
What amount of the excess reagent remains when 0.30 mol NH3 reacts with 0.40 mol O2 to produce NO and H2O? 4NH3 +502 + 4NO + 6H20
(A) 0.10 mol NH,
(B) 0.10 mol
(C) 0.025 mol NH
(D) 0.025 mol O2

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

Chemistry, 22.06.2019 20:00
If one fission reaction of a uranium-235 atom produced two neutrons, how many neutrons would be released if the chain reaction occurred three more times?
Answers: 1
You know the right answer?
What amount of the excess reagent remains when 0.30 mol NH3 reacts with 0.40 mol O2 to produce NO an...
Questions


Biology, 22.06.2019 17:30


Mathematics, 22.06.2019 17:30

Mathematics, 22.06.2019 17:30


Mathematics, 22.06.2019 17:30

Mathematics, 22.06.2019 17:30

History, 22.06.2019 17:30


Business, 22.06.2019 17:30



Mathematics, 22.06.2019 17:30





Mathematics, 22.06.2019 17:30

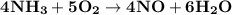
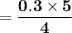
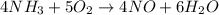
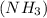 reacts with = 5 moles of oxygen
reacts with = 5 moles of oxygen 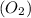
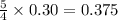 moles of oxygen
moles of oxygen 

