
Chemistry, 13.12.2020 04:40 jadeochoa4466
For the following reaction, calculate how many moles of each product are formed when 4.05 g of water is used. 2H20 →2H2 + O2

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 16:00
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 22.06.2019 22:30
The diagram shows the relationship between scientific disciplines.the names of some scientific disciplines have been removed from the boxes. which scientific discipline belongs in the blue box? a.physics b.biology c.chemistry d.metallurgy
Answers: 2
You know the right answer?
For the following reaction, calculate how many moles of each product are formed when 4.05 g of water...
Questions


Mathematics, 19.03.2021 20:50

Mathematics, 19.03.2021 20:50

Mathematics, 19.03.2021 20:50

Mathematics, 19.03.2021 20:50

Mathematics, 19.03.2021 20:50



Mathematics, 19.03.2021 20:50

History, 19.03.2021 20:50





Mathematics, 19.03.2021 20:50


English, 19.03.2021 20:50



Mathematics, 19.03.2021 20:50

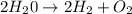
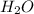 is used to form 2 moles of
is used to form 2 moles of 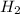 and 1 mole of
and 1 mole of 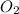
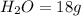 , 1 mole of
, 1 mole of 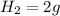 , and 1 mole of
, and 1 mole of 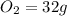 .
.

