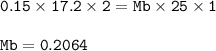Chemistry, 13.12.2020 23:20 bekahmc1p6k6vj
A student used a pipette to add 25.0 cm of KOH of unknown concentration to
a conical flask.
The student carried out a titration experiment to find the volume of 0.150 mol/dmº
H2SO4 needed to neutralise the KOH.
The student found that, on average, 17.20 cm of the H2SO4 solution was
required for neutralisation.
Calculate the concentration of the KOH solution.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:30
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1

Chemistry, 22.06.2019 16:00
Which process transfers heat from inside earth to its surface? convection currents in mantle pulling away of tectonic plates drawing in of tectonic plates convection currents in crust
Answers: 1


Chemistry, 23.06.2019 07:00
The following transition occurs at a molecular level for a substance. what transition corresponds to this change in microscopic structure? the carbon dioxide molecules on the left are in a regular, tightly packed pattern. after heating, it becomes much lower density. a. melting b. boiling c. sublimation d. freezing
Answers: 1
You know the right answer?
A student used a pipette to add 25.0 cm of KOH of unknown concentration to
a conical flask.
Th...
Th...
Questions



Mathematics, 16.10.2020 05:01



Physics, 16.10.2020 05:01

Chemistry, 16.10.2020 05:01

Mathematics, 16.10.2020 05:01

Mathematics, 16.10.2020 05:01

History, 16.10.2020 05:01


History, 16.10.2020 05:01


Chemistry, 16.10.2020 05:01



English, 16.10.2020 05:01

Mathematics, 16.10.2020 05:01


Mathematics, 16.10.2020 05:01