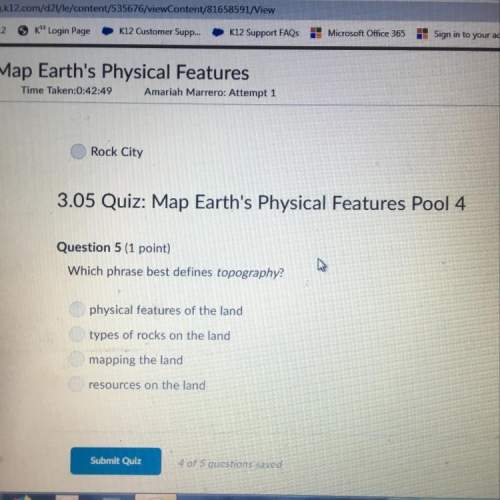
Chemistry, 14.12.2020 07:20 oliviaberta91
State Graham's law of diffusion. A vessel of volume 100ml
contains 10% O2 and 90% unknown gas. The gases diffuse in 86
sec. through a small hole of the vessel. If pure oxygen under the
same condition diffuses in 75 sec. Find the molecular mass of
unknown gas.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:30
What volume of a 2.00 m stock solution of naoh is needed to prepare 150. ml of 0.40 m solution?
Answers: 2

Chemistry, 22.06.2019 02:00
What is the volume occupied by 10.0 dm3 of gas at standard pressure after it has been compressedat constant temputure to 500.0 kpa?
Answers: 1

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

You know the right answer?
State Graham's law of diffusion. A vessel of volume 100ml
contains 10% O2 and 90% unknown gas. The...
Questions

History, 09.04.2020 10:23

Mathematics, 09.04.2020 10:23

Chemistry, 09.04.2020 10:23

Mathematics, 09.04.2020 10:23

Mathematics, 09.04.2020 10:23



Mathematics, 09.04.2020 10:23

Mathematics, 09.04.2020 10:23

English, 09.04.2020 10:24

Mathematics, 09.04.2020 10:24

Mathematics, 09.04.2020 10:24



Mathematics, 09.04.2020 10:24


Mathematics, 09.04.2020 10:24

Mathematics, 09.04.2020 10:24


Social Studies, 09.04.2020 10:24