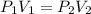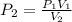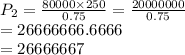The pressure inside of a sealed syringe of hydrogen gas is 80.0KPa with a volume of 250 Liters. What is the pressure inside
of the syringe after the plunger is pulled back far enough to make final volume of the syringe 0.750 Liters? (assume the
temperature and amount of gas in the container are held constant)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3


Chemistry, 23.06.2019 01:30
What is the importance of interlocking the fingers and rubbing while washing hands? the palms are the dirtiest parts of the hands. the spaces between the fingers get washed. the backs of the hands get washed. the fingernails are the dirtiest parts of the hands
Answers: 1

Chemistry, 23.06.2019 02:30
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3
You know the right answer?
The pressure inside of a sealed syringe of hydrogen gas is 80.0KPa with a volume of 250 Liters. What...
Questions

English, 13.10.2020 14:01

Arts, 13.10.2020 14:01



Health, 13.10.2020 14:01

Health, 13.10.2020 14:01

Social Studies, 13.10.2020 14:01



English, 13.10.2020 14:01

History, 13.10.2020 14:01

Arts, 13.10.2020 14:01

Mathematics, 13.10.2020 14:01

Biology, 13.10.2020 14:01


Mathematics, 13.10.2020 14:01


Mathematics, 13.10.2020 14:01

Mathematics, 13.10.2020 14:01

Mathematics, 13.10.2020 14:01