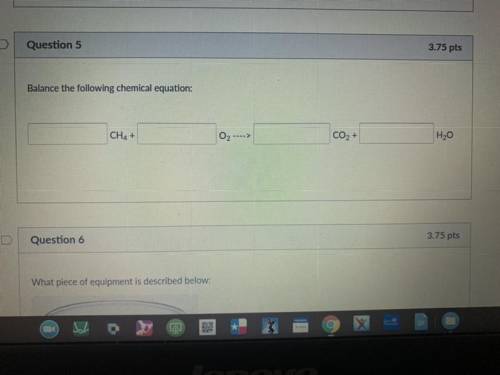
Balance the following chemical equation:
———CH4+———O2- - -> ———CO2+———H2O
...

Chemistry, 17.12.2020 22:50 lashondrascott
Balance the following chemical equation:
———CH4+———O2- - -> ———CO2+———H2O

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 11:00
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced.be sure your answer has the correct number of significant digits.
Answers: 2

Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3

Chemistry, 23.06.2019 00:00
How many moles of water are created if 3 moles of hydrogen react completely with excess oxygen?
Answers: 3
You know the right answer?
Questions


Social Studies, 21.01.2021 18:50

Mathematics, 21.01.2021 18:50

English, 21.01.2021 18:50

English, 21.01.2021 18:50

Mathematics, 21.01.2021 18:50


Mathematics, 21.01.2021 18:50

Mathematics, 21.01.2021 18:50


History, 21.01.2021 18:50




English, 21.01.2021 18:50

Mathematics, 21.01.2021 18:50

Chemistry, 21.01.2021 18:50


Mathematics, 21.01.2021 18:50

Mathematics, 21.01.2021 18:50



