
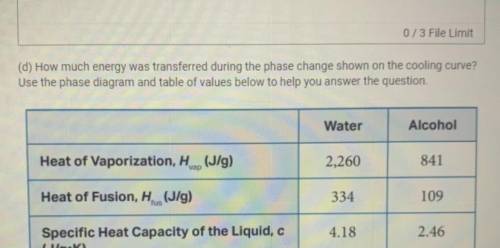
(d) How much energy was transferred during the phase change shown on the cooling curve?
Use the phase diagram and table of values below to help you answer the question.
Water
Alcohol
Heat of Vaporization, Hvap (J/g)
2,260
841
Heat of Fusion, H. (J/g)
334
fus
109
4.18
Specific Heat Capacity of the Liquid, c
(J/g. K)
2.46

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 23.06.2019 00:30
On the periodic table, elements are arranged by which of the following. a. mass numbers. b. increasing atomic number. c. alphabetical order. or d. density
Answers: 1

Chemistry, 23.06.2019 01:30
Will a solution form when the solvent and solute are both nonpolar? a. not likely b. never c. most likely
Answers: 1
You know the right answer?
(d) How much energy was transferred during the phase change shown on the cooling curve?
Use the pha...
Questions

Business, 10.12.2019 18:31



Mathematics, 10.12.2019 18:31







Computers and Technology, 10.12.2019 18:31


Social Studies, 10.12.2019 18:31

Mathematics, 10.12.2019 18:31

History, 10.12.2019 18:31

Mathematics, 10.12.2019 18:31







