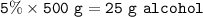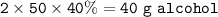Chemistry, 21.12.2020 14:00 expeditionofsin
Mr. Costas drank a bottle of beer (500g) containing 5% w / w alcohol, while Mr. George drank two drinks (50g each) vodka containing 40% w / w alcohol. How much alcohol did each of them consume? Which of the two drank the most alcohol?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2

You know the right answer?
Mr. Costas drank a bottle of beer (500g) containing 5% w / w alcohol, while Mr. George drank two dri...
Questions



Mathematics, 23.09.2019 03:30

Mathematics, 23.09.2019 03:30

Mathematics, 23.09.2019 03:30

History, 23.09.2019 03:30


Chemistry, 23.09.2019 03:30

Physics, 23.09.2019 03:40





World Languages, 23.09.2019 03:50



Mathematics, 23.09.2019 03:50

History, 23.09.2019 03:50

Mathematics, 23.09.2019 03:50

Mathematics, 23.09.2019 03:50