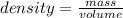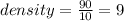Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 12:00
An atom's configuration based on its number of electrons ends at 3p4. another atom has seven more electrons. starting at 3p, what is the remaining configuration? 3p63d34s2 3p43d54s2 3p64s23d3 3p44s23d
Answers: 3

Chemistry, 22.06.2019 14:30
Chemistry worksheet - i am not sure what they are asking for exactly?
Answers: 1
You know the right answer?
A 10.0 cm3 sample of copper has a mass of 90 g. What is the density of copper?...
Questions

Law, 12.04.2021 14:00


Mathematics, 12.04.2021 14:00

Mathematics, 12.04.2021 14:00

Mathematics, 12.04.2021 14:00


Mathematics, 12.04.2021 14:00

Mathematics, 12.04.2021 14:00

Social Studies, 12.04.2021 14:00




Business, 12.04.2021 14:00


History, 12.04.2021 14:00


Advanced Placement (AP), 12.04.2021 14:00

Mathematics, 12.04.2021 14:00

Computers and Technology, 12.04.2021 14:00