Chemistry, 26.12.2020 21:40 ashleyrover11
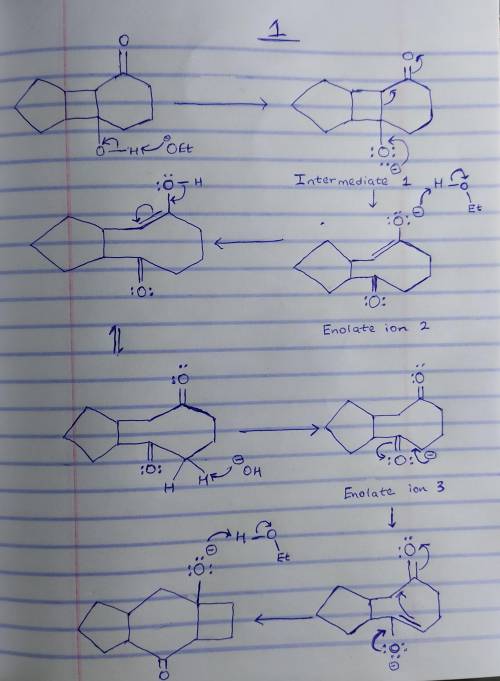
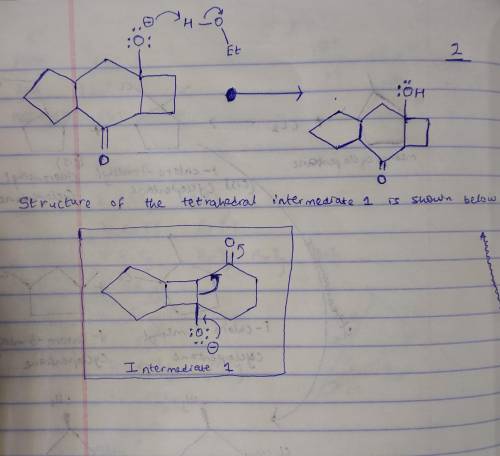
This base-catalyzed isomerization involves the following steps: 1. Deprotonation to form tetrahedral intermediate 1; 2. Collapse of the tetrahedral intermediate and bond cleavage to form enolate ion 2; 3. Proton transfer to form enolate ion 3; 4. Intramolecular aldol reaction to form tetrahedral intermediate 4. Write out the reaction on a separate sheet of paper, and then draw the structure of tetrahedral intermediate 1.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:40
Apiece of copper has a temperature of 75.6 0c. when the metal is placed in 100.0 grams of water at 19.1 0c, the temperature rises by 5.5 0c. what is the mass of the metal?
Answers: 1

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 22.06.2019 14:00
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3
You know the right answer?
This base-catalyzed isomerization involves the following steps: 1. Deprotonation to form tetrahedral...
Questions


Spanish, 15.10.2019 04:00



Health, 15.10.2019 04:00

English, 15.10.2019 04:00



Mathematics, 15.10.2019 04:00

Biology, 15.10.2019 04:00


History, 15.10.2019 04:00


History, 15.10.2019 04:00


English, 15.10.2019 04:00

Geography, 15.10.2019 04:00