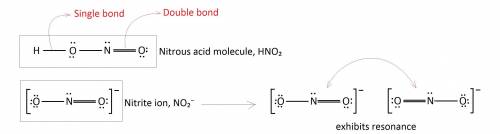
Incomplete Lewis structures for the nitrous acid molecule, HNO2, and the nitrite ion, NO2-, are shown here. (a) Complete each Lewis structure by adding electron pairs as needed. (b) Is the formal charge on N the same or different in these two species? (c) Would either HNO2 or NO2- be expected to exhibit resonance?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:50
Calculate the number of molecules present in 0.750 mol of mgo.
Answers: 3

Chemistry, 22.06.2019 07:30
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1
You know the right answer?
Incomplete Lewis structures for the nitrous acid molecule, HNO2, and the nitrite ion, NO2-, are show...
Questions

Mathematics, 04.04.2021 20:10

History, 04.04.2021 20:10

English, 04.04.2021 20:10

Mathematics, 04.04.2021 20:10

Mathematics, 04.04.2021 20:20

History, 04.04.2021 20:20




Biology, 04.04.2021 20:20

History, 04.04.2021 20:20

Mathematics, 04.04.2021 20:20

English, 04.04.2021 20:20

Health, 04.04.2021 20:20




Mathematics, 04.04.2021 20:20


Chemistry, 04.04.2021 20:20