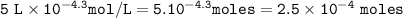Additional Practice Questions
5. If you had a pH of 4.30 in a 5.00 L sample, how many
moles o...

Chemistry, 06.01.2021 03:40 wwwcarolynzouowficz
Additional Practice Questions
5. If you had a pH of 4.30 in a 5.00 L sample, how many
moles of hydronium do you have?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:10
Nitrogen (n), phosphorus (p), and potassium (k) are the main nutrients in plant fertilizers. according to an industry convention, the numbers on the label refer to the mass percents of n, p2o5, and k2o, in that order. calculate the n: p: k ratio of a 30: 10: 10 fertilizer in terms of moles of each element, and express it as x: y: 1.0.
Answers: 1

Chemistry, 22.06.2019 15:10
The ozone molecule o3 has a permanent dipole moment of 1.8×10−30 cm. although the molecule is very slightly bent-which is why it has a dipole moment-it can be modeled as a uniform rod of length 2.5×10−10 m with the dipole moment perpendicular to the axis of the rod. suppose an ozone molecule is in a 8000 n/c uniform electric field. in equilibrium, the dipole moment is aligned with the electric field. but if the molecule is rotated by a small angle and released, it will oscillate back and forth in simple harmonic motion.what is the frequency f of oscillation?
Answers: 2

Chemistry, 22.06.2019 16:00
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1

You know the right answer?
Questions


Mathematics, 26.11.2021 20:10

Mathematics, 26.11.2021 20:10

English, 26.11.2021 20:10


Mathematics, 26.11.2021 20:10



English, 26.11.2021 20:10


Biology, 26.11.2021 20:10


Social Studies, 26.11.2021 20:10

Social Studies, 26.11.2021 20:10


Mathematics, 26.11.2021 20:10



Chemistry, 26.11.2021 20:10

![\tt [H_3O^+]=10^{-4.3}~M=10^{-4.3}mol/L](/tpl/images/1015/5497/88b81.png)