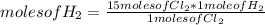Chemistry, 06.01.2021 17:30 trizthagod49
How many moles of hydrogen gas(H2) are needed to react with 15 moles of chlorine gas

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 05:40
Why is any chemical reaction always balanced? give reasons and explain the easiest way to solve the balancing problems in chemical equations with stoichiometric coefficients upto 20 as hit and trial doesn't always work. give full reasoning
Answers: 1

You know the right answer?
How many moles of hydrogen gas(H2) are needed to react with 15 moles of chlorine gas...
Questions

Mathematics, 11.10.2020 14:01


English, 11.10.2020 14:01





English, 11.10.2020 14:01


Mathematics, 11.10.2020 14:01

Advanced Placement (AP), 11.10.2020 14:01



Mathematics, 11.10.2020 14:01



Physics, 11.10.2020 14:01

Mathematics, 11.10.2020 14:01

English, 11.10.2020 14:01

Biology, 11.10.2020 14:01