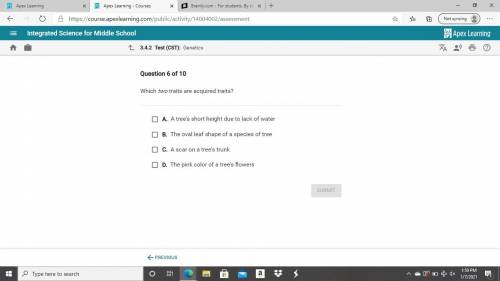
Please help me its science class
...

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 03:50
How many liters of oxygen gas, at standardtemperature and pressure, will react with 35.8 grams ofiron metal? 4 fe (s) + 3 o2 (g) → 2 fe2o3 (s)
Answers: 3
You know the right answer?
Questions

Mathematics, 06.04.2021 01:50

Mathematics, 06.04.2021 01:50

Mathematics, 06.04.2021 01:50

Mathematics, 06.04.2021 01:50

Physics, 06.04.2021 01:50

Mathematics, 06.04.2021 01:50


Computers and Technology, 06.04.2021 01:50


Mathematics, 06.04.2021 01:50







English, 06.04.2021 02:00

Mathematics, 06.04.2021 02:00


Physics, 06.04.2021 02:00