
Chemistry, 07.01.2021 23:50 gshreya2005
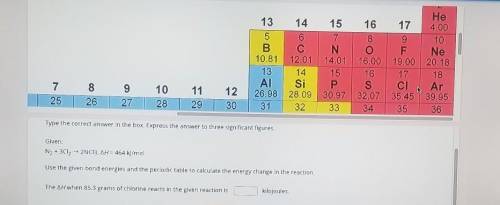
Type the correct answer in the box, Express the answer to three significant figures. Given: N2 + 3Cl2 + 2NC3, AH = 464 kJ/mol Use the given bond energies and the periodic table to calculate the energy change in the reaction. The AH when 85.3 grams of chlorine reacts in the given reaction is kilojoules.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2

Chemistry, 22.06.2019 13:00
In a copper wire, a temperature increase is the result of which of the following
Answers: 1
You know the right answer?
Type the correct answer in the box, Express the answer to three significant figures. Given: N2 + 3Cl...
Questions


History, 19.09.2019 11:00

Computers and Technology, 19.09.2019 11:00



Mathematics, 19.09.2019 11:00


Biology, 19.09.2019 11:00



Computers and Technology, 19.09.2019 11:00


Chemistry, 19.09.2019 11:00


Physics, 19.09.2019 11:00

Geography, 19.09.2019 11:00

History, 19.09.2019 11:00


English, 19.09.2019 11:00

Mathematics, 19.09.2019 11:00



