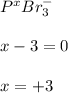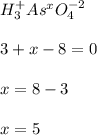Chemistry, 08.01.2021 03:40 kierraware04
g Determine the oxidation state for each of the elements below. ... The oxidation state of ... phosphorus ... in ... phosphorous tribromide PBr3 ... is ... . The oxidation state of nitrogen in nitrogen gas N2 is . The oxidation state of arsenic in arsenic acid H3AsO4 is .

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:30
**40** points asapessay questions (10 points possible) clear image of next, create your own scenario. it can be one of your own real experiences or one you make up. use imagery in your writing to give your instructor a the setting and an action taking pace in your writing explain the structure and functions of the skin at work in your scenario. !
Answers: 3

Chemistry, 21.06.2019 22:50
Blank allows you to do calculations for situations in which only the amount of gas is constant a)boyle's law b)combined gas law c)ideal gas law d)dalton's law
Answers: 1

Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2

You know the right answer?
g Determine the oxidation state for each of the elements below. ... The oxidation state of ... phosp...
Questions

Social Studies, 30.05.2020 21:01






English, 30.05.2020 21:01

Mathematics, 30.05.2020 21:01

Mathematics, 30.05.2020 21:01



Mathematics, 30.05.2020 21:01


English, 30.05.2020 21:01

Mathematics, 30.05.2020 21:01




Mathematics, 30.05.2020 21:01