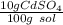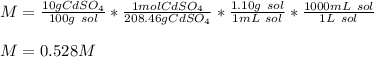Chemistry, 08.01.2021 17:10 danteyoungblood7
What is the molarity of a solution of 10% by mass cadmium sulfate, CdSO4 (molar mass = 208.46 g/mol) by mass? The density of the solution is 1.10 g/mL.
a) 0.528 M
b) 0.436 M
c) 0.479 M
d) 0.048 M
e) 22.9 M

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:10
Harvey mixes two liquids. which observation of the new mixture most likely indicates a precipitate is forming?
Answers: 2

Chemistry, 22.06.2019 06:00
How much would the freezing point of water decrease if 4 mol of sugar were added to 1 kg of water(k=1.86 c/mol/kg for water and i=1 for sugar
Answers: 1

Chemistry, 22.06.2019 16:00
About 3% of the water on earth is freshest. only about 40% of that freshwater is available for human use. why is so much freshwater unavailable for human use?
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3
You know the right answer?
What is the molarity of a solution of 10% by mass cadmium sulfate, CdSO4 (molar mass = 208.46 g/mol)...
Questions






Chemistry, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01

English, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01


Mathematics, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01


Chemistry, 20.10.2020 21:01

History, 20.10.2020 21:01



English, 20.10.2020 21:01

Biology, 20.10.2020 21:01

Health, 20.10.2020 21:01