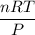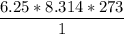Chemistry, 10.01.2021 18:30 kaylaamberd
En un matraz, disponemos de 100 g de gas oxígeno que se encuentran a 1 at de presión y 273 K de temperatura. Calcular : a) el número de moles de gas oxígeno contenidos en el matraz ; b) el número de moléculas de oxígeno ; c) el número de átomos de oxígeno ; d) el volumen ocupado por el oxígeno. Masa atómica del oxígeno = 16.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:10
How do forces between particles in gases compare to forces in the other states of matter? o a. the forces in gases are stronger than forces in solids but weaker than forces in liquids. o b. the forces in gases are weaker than forces in solids but stronger than forces in liquids. o c. the forces in gases are weaker than forces in solids and liquids. o d. the forces in gases are stronger than forces in solids and liquids. submit
Answers: 1

Chemistry, 21.06.2019 22:50
Blank allows you to do calculations for situations in which only the amount of gas is constant a)boyle's law b)combined gas law c)ideal gas law d)dalton's law
Answers: 1

Chemistry, 23.06.2019 00:00
Predict the relative bond lengths of the three carbon-oxygen bonds in the carbonate ion (co2−3). what would you expect the charge to be on each oxygen? match the words in the left column to the appropriate blanks in the sentences on the right. make certain each sentence is complete before submitting your answer.
Answers: 3

Chemistry, 23.06.2019 05:40
The independent variable in an experiment will be the variable that you o a) change ob) hold constant ng c) observe for changes
Answers: 2
You know the right answer?
En un matraz, disponemos de 100 g de gas oxígeno que se encuentran a 1 at de presión y 273 K de temp...
Questions

Law, 07.12.2020 01:50

Mathematics, 07.12.2020 01:50


Mathematics, 07.12.2020 01:50


Social Studies, 07.12.2020 01:50


Biology, 07.12.2020 01:50

Mathematics, 07.12.2020 01:50

Business, 07.12.2020 01:50

Mathematics, 07.12.2020 01:50


Geography, 07.12.2020 01:50

Mathematics, 07.12.2020 01:50

Health, 07.12.2020 01:50

Mathematics, 07.12.2020 01:50

Law, 07.12.2020 01:50

Mathematics, 07.12.2020 01:50