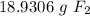Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3
You know the right answer?
Find the mass in grams of 3.00 x 1023 molecules of F2...
Questions


Mathematics, 11.06.2020 16:57

Mathematics, 11.06.2020 16:57

History, 11.06.2020 16:57


Computers and Technology, 11.06.2020 16:57



Social Studies, 11.06.2020 16:57






Computers and Technology, 11.06.2020 16:57




History, 11.06.2020 16:57

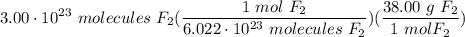 Multiply:
Multiply: