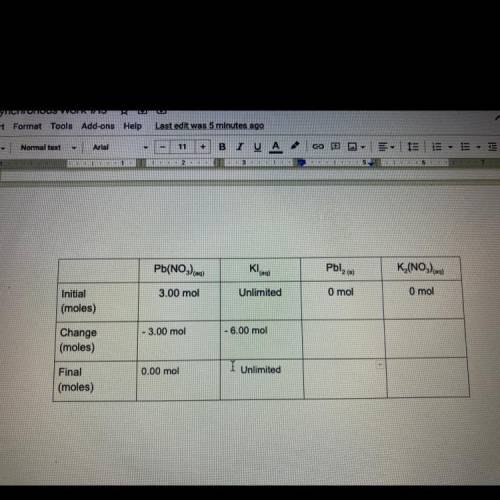
Task 4
3.00 moles of lead nitrate and excess potassium iodidq are combined in water to
form l...

Task 4
3.00 moles of lead nitrate and excess potassium iodidq are combined in water to
form lead iodide and potassium nitrate. The reaction is described by the following
chemical equation:
Pb(NO3) eq) + 2Kleg) Pblzco + K (NO)
Fill out the following table to find out how many moles of products are formed,
and how many of the reactants remain after the reaction has taken place. The boxes in
the final row are all mole to mole conversions, and can be found by using your molar
ratios and the moles of lead nitrate at the beginning of the reaction. It is your choice
how you will arrange these two numbers. If this problem becomes confusing, look back
to task 3, It's the same problem!

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 13:30
Ants live on acacia trees in south america. the ants feed on sugars secreted by the trees. the trees provide room for the ants to live. the ants sting any other insect or animal that comes to eat the trees. what type of relationship is this?
Answers: 1

Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

Chemistry, 22.06.2019 14:20
7. in the cycle, a virus integrates its dna into the host's dna, and its dna is replicated when the host dna is replicated. a. infectious b. retroviral c. lysogenic d.lytic
Answers: 1

Chemistry, 22.06.2019 23:00
Which type of intermolecular attractions holds ammonia molecules together with other ammonia molecules?
Answers: 3
You know the right answer?
Questions


Mathematics, 15.10.2020 21:01







Biology, 15.10.2020 21:01

English, 15.10.2020 21:01



Mathematics, 15.10.2020 21:01

English, 15.10.2020 21:01

Mathematics, 15.10.2020 21:01

Mathematics, 15.10.2020 21:01



Chemistry, 15.10.2020 21:01

History, 15.10.2020 21:01



