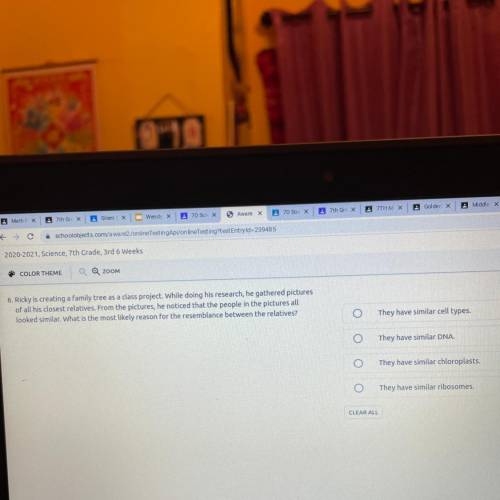
Help me please I really need help and answer only if u know the answer
...


Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3


Chemistry, 23.06.2019 09:00
Describe the process that was used in this lab to create magnesium oxide, specifically identifying the type of chemical reaction. explain why the product had a higher mass than the reactant, and how this relates to conservation of matter.
Answers: 2

You know the right answer?
Questions







Mathematics, 30.03.2021 03:40



Mathematics, 30.03.2021 03:40


Mathematics, 30.03.2021 03:40

Mathematics, 30.03.2021 03:40


World Languages, 30.03.2021 03:40

Mathematics, 30.03.2021 03:40

Mathematics, 30.03.2021 03:40



Computers and Technology, 30.03.2021 03:40