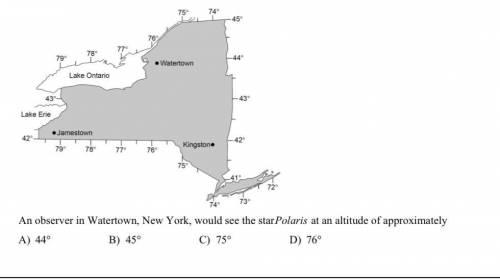
Please explain why the answer is correct and why the others aren’t.
Thank you!!
...

Chemistry, 18.01.2021 01:00 ymlantigua21
Please explain why the answer is correct and why the others aren’t.
Thank you!!

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 13:00
Asolution has a ca2+ concentration of 0.049 m and an f- concentration is 0.147 m at equilibrium. the value of ksp for caf2 at 25°c is 4.0 x 10-11. will this solution form a precipitate? yes no
Answers: 3

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2
You know the right answer?
Questions

Mathematics, 14.01.2021 04:10





English, 14.01.2021 04:10

English, 14.01.2021 04:10

Mathematics, 14.01.2021 04:10


Mathematics, 14.01.2021 04:10

Mathematics, 14.01.2021 04:10

Mathematics, 14.01.2021 04:10

Biology, 14.01.2021 04:10


Geography, 14.01.2021 04:20

Mathematics, 14.01.2021 04:20

Advanced Placement (AP), 14.01.2021 04:20


Mathematics, 14.01.2021 04:20

Mathematics, 14.01.2021 04:20



