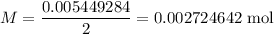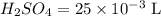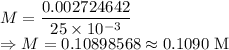Chemistry, 18.01.2021 14:00 AgarioEdit
A 25.00 mL sample of an H2SO4 solution of unknown concentration is titrated with a 0.1322 M KOH solution. A volume of 41.22 mL of KOH is required to reach the equivalence point. What is the concentration of the unknown H2SO4 solution? Express your answer in molarity to four significant figures.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Si una estrella no tiene paralaje medible, ¿qué puedes inferir?
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1
You know the right answer?
A 25.00 mL sample of an H2SO4 solution of unknown concentration is titrated with a 0.1322 M KOH solu...
Questions



Mathematics, 20.07.2019 19:00


Chemistry, 20.07.2019 19:00

Mathematics, 20.07.2019 19:00


English, 20.07.2019 19:00

Mathematics, 20.07.2019 19:00

Mathematics, 20.07.2019 19:00

Mathematics, 20.07.2019 19:00

Mathematics, 20.07.2019 19:00

English, 20.07.2019 19:00




Geography, 20.07.2019 19:00



Mathematics, 20.07.2019 19:00

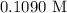
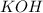 = 41.22 mL
= 41.22 mL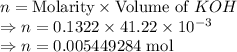
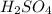
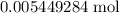 of
of