
Chemistry, 18.01.2021 16:50 dragongacha777
PLEASE HELP ME
1. What is the molality of a solution made up of 43.6 mol of CaCl2 dissolved by 13.5 kg of water?
2. How many moles of Na2CO3 are required to create 9.54 liters of a 3.4 M solution?
3. Which of these actions can result in decreasing the molarity of a solution?
Select all that apply.
A
adding solute
B
adding solvent
C
removing solute
D
removing solvent
Thanks Y'all:)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Maria wants to determine which type of disinfectant kills the most bacteria. which of the following is the best way for maria to determine this? a. ask ten different companies that make disinfectants which type is best. b. put the same amount and species of bacteria on ten identical plates, and add ten different kinds of disinfectant to each plate. c. interview ten different people to determine which type of disinfectant they prefer. d. put the same amount and species of bacteria on ten identical plates, and add a different disinfectant to each plate.
Answers: 1

Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1

Chemistry, 22.06.2019 22:10
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1

Chemistry, 23.06.2019 02:00
Why does ammonia, nh3, behave as a base when it reacts with an acid? z
Answers: 2
You know the right answer?
PLEASE HELP ME
1. What is the molality of a solution made up of 43.6 mol of CaCl2 dissolved by 13.5...
Questions


Mathematics, 15.11.2021 01:00


English, 15.11.2021 01:00



Mathematics, 15.11.2021 01:00

Mathematics, 15.11.2021 01:00

Mathematics, 15.11.2021 01:00

Mathematics, 15.11.2021 01:00

Computers and Technology, 15.11.2021 01:00

Computers and Technology, 15.11.2021 01:00

Chemistry, 15.11.2021 01:00

History, 15.11.2021 01:00

Mathematics, 15.11.2021 01:00

Computers and Technology, 15.11.2021 01:00



Mathematics, 15.11.2021 01:00

Biology, 15.11.2021 01:00

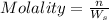
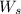 = weight of solvent
= weight of solvent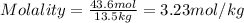
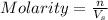
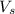 = volume of solution in L
= volume of solution in L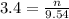
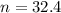
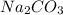 is 32.4
is 32.4 

