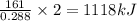Chemistry, 18.01.2021 21:30 jordandabrat
The reaction of hydrogen sulfide(g) with oxygen(g) to form water(l) and sulfur dioxide(g) proceeds as follows: 2H2S(g) 3O2(g)2H2O(l) 2SO2(g) When 9.82 g H2S(g) reacts with sufficient O2(g), 161 kJ is evolved. Calculate the value of rH for the chemical equation given. kJ/mol

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2

Chemistry, 23.06.2019 01:00
The time that is taken by neptune once around the sun is called
Answers: 1

Chemistry, 23.06.2019 12:50
What is the relative mass of an electron? a) 1/1840 the mass of a neutron + proton b) 1/1840 the mass of an alpha particle c) 1/1840 the mass of a c-12 atom d) 1/1840 the mass of a hydrogen atom
Answers: 3

Chemistry, 23.06.2019 15:50
Many radioactive atoms that have large masses undergo radioactive decay by releasing a particle that is identical to a helium-4 nucleus. what changes in the original atom are expected as a result of this natural phenomenon? the atomic number and the mass number will decrease. the atomic number and the mass number will increase. the atomic number will increase, and the mass number will decrease. the atomic number will decrease, and the mass number will increase.
Answers: 2
You know the right answer?
The reaction of hydrogen sulfide(g) with oxygen(g) to form water(l) and sulfur dioxide(g) proceeds a...
Questions


Mathematics, 08.03.2021 21:30



Social Studies, 08.03.2021 21:30


Mathematics, 08.03.2021 21:30

Chemistry, 08.03.2021 21:30

Health, 08.03.2021 21:30



Mathematics, 08.03.2021 21:30



Business, 08.03.2021 21:30


Chemistry, 08.03.2021 21:30


Mathematics, 08.03.2021 21:30

Mathematics, 08.03.2021 21:30

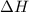 is 1118 .
is 1118 .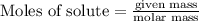
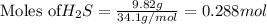
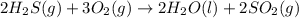
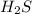 is reacted = 161 kJ
is reacted = 161 kJ