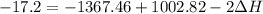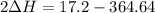Chemistry, 19.01.2021 19:40 supergraciepie
A scientist measures the standard enthalpy change for the following reaction to be -17.2 kJ : Ca(OH)2(aq) 2 HCl(aq)CaCl2(s) 2 H2O(l) Based on this value and the standard enthalpies of formation for the other substances, the standard enthalpy of formation of HCl(aq) is kJ/mol.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

Chemistry, 22.06.2019 12:30
In france, grapes are 1.95 euros per kilogram. what is the cost of grapes, in dollars per pound, if the exchange rate is 1.14 dollars/euro? (2.6)
Answers: 3

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2
You know the right answer?
A scientist measures the standard enthalpy change for the following reaction to be -17.2 kJ : Ca(OH)...
Questions






Computers and Technology, 06.12.2019 02:31


Social Studies, 06.12.2019 02:31






Computers and Technology, 06.12.2019 02:31






 kJ/mol
kJ/mol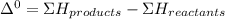
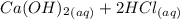 →
→ 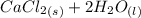
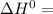 -1002.82 kJ/mol
-1002.82 kJ/mol![-17.2=[-795.8+2(285.85)]-[-1002.82+2\Delta H]](/tpl/images/1045/5683/27b56.png)