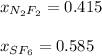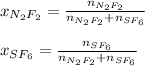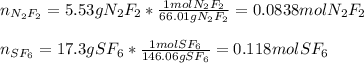Chemistry, 21.01.2021 23:00 darknessmidnight207
A 8.00 L tank at 26.9 C is filled with 5.53 g of dinitrogen difluoride gas and 17.3 g of sulfur hexafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction of each gas. Round each of your answers to significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 06:30
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1

Chemistry, 22.06.2019 21:30
How many oxygen atoms are there in 3.15 moles of hcl manganese (iv) oxide, mno2
Answers: 2

You know the right answer?
A 8.00 L tank at 26.9 C is filled with 5.53 g of dinitrogen difluoride gas and 17.3 g of sulfur hexa...
Questions


Geography, 11.01.2020 03:31

Mathematics, 11.01.2020 03:31

Social Studies, 11.01.2020 03:31


History, 11.01.2020 03:31




Mathematics, 11.01.2020 03:31





History, 11.01.2020 03:31

Computers and Technology, 11.01.2020 03:31

Chemistry, 11.01.2020 03:31


Mathematics, 11.01.2020 03:31