

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 22.06.2019 21:30
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1

Chemistry, 22.06.2019 22:00
The diagrams to the right show the distribution and arrangement of gas particles in two different containers. according to kinetic-molecular theory, which of the following statements is true? check all that apply. if the temperatures of both containers are equal, container a has greater pressure than container b. if the volume of container a decreased, its pressure would decrease. if the pressure in both containers is equal, container a has a lower temperature than container b.
Answers: 2

Chemistry, 22.06.2019 23:00
What extra step distinguishes fermentation from glycolysis
Answers: 1
You know the right answer?
The density of whole milk 1.04 g per mL. What is the volume (in quarts) of 18.5 pounds of whole milk...
Questions

Social Studies, 02.09.2019 02:10

Chemistry, 02.09.2019 02:10

Mathematics, 02.09.2019 02:10



Mathematics, 02.09.2019 02:10

English, 02.09.2019 02:10



History, 02.09.2019 02:10



Mathematics, 02.09.2019 02:10

Mathematics, 02.09.2019 02:10




Mathematics, 02.09.2019 02:10

Biology, 02.09.2019 02:10

Mathematics, 02.09.2019 02:10

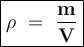 .
.

