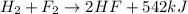Chemistry, 25.01.2021 20:40 sandersasia2
When H2(g) reacts with F2(g) to form HF(g) , 542 kJ of energy are evolved for each mole of H2(g) that reacts. Write a balanced thermochemical equation for the reaction with an energy term in kJ as part of the equation. Note that the answer box for the energy term is case sensitive. Use the SMALLEST INTEGER coefficients possible and put the energy term (including the units) in the last box on the appropriate side of the equation. If a box is not needed, leave it blank.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Diffraction is when light is bent around obstructions. which of the these observation about clouds would indicate diffraction? a) after rain storms, you can sometimes see rainbows. b) clouds are white or gray and cannot be seen through. c) on a cloudy day, the temperature tends to be cooler than a sunny day. d) the edges of dark clouds appear lighter. this
Answers: 3

Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1


Chemistry, 23.06.2019 03:00
Abaker touches a pie right after taking it out of the oven. which statement best explains why the pie feels hot?
Answers: 1
You know the right answer?
When H2(g) reacts with F2(g) to form HF(g) , 542 kJ of energy are evolved for each mole of H2(g) tha...
Questions


Mathematics, 26.06.2019 11:00

Mathematics, 26.06.2019 11:00


Mathematics, 26.06.2019 11:00

Mathematics, 26.06.2019 11:00

Biology, 26.06.2019 11:00


Business, 26.06.2019 11:00









English, 26.06.2019 11:00