Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 22.06.2019 22:10
Which aqueous solution of ki freezes at the lowest temperature? 1) 1 mol of ki in 500. g of water 2) 2 mol of ki in 500. g of water 3) 1 mol of ki in 1000. g of water 4) 2 mol of ki in 1000. g of water
Answers: 3

Chemistry, 23.06.2019 03:30
How many grams of sodium chloride are in 250ml of a 2.5m naci solution
Answers: 1
You know the right answer?
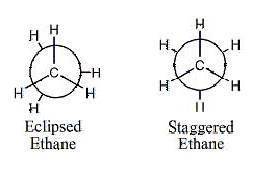
Part I: SP3 Hybridization: B: Ethane Distance between C-C in ethane: Distance between H atoms in ec...
Questions

Mathematics, 02.03.2021 06:00

History, 02.03.2021 06:00

Social Studies, 02.03.2021 06:00

Social Studies, 02.03.2021 06:00

Mathematics, 02.03.2021 06:00

Mathematics, 02.03.2021 06:00






Mathematics, 02.03.2021 06:00

Chemistry, 02.03.2021 06:00

Mathematics, 02.03.2021 06:00





Spanish, 02.03.2021 06:00