L
10) A pure sample of an element was vaporized and injected into a mass spectrometer
and the...

Chemistry, 25.01.2021 21:40 stacywashburnstu
L
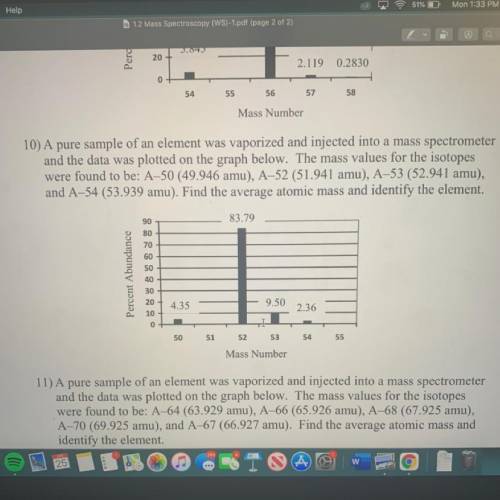
10) A pure sample of an element was vaporized and injected into a mass spectrometer
and the data was plotted on the graph below. The mass values for the isotopes
were found to be: A–50 (49.946 amu), A-52 (51.941 amu), A-53 (52.941 amu),
and A-54 (53.939 amu). Find the average atomic mass and identify the element.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
In numbering carbon atoms in the parent chain of a hydrocarbon, why would you number from right to left, rather than left to right
Answers: 1

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1


Chemistry, 22.06.2019 17:10
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1
You know the right answer?
Questions


Social Studies, 27.11.2020 22:50

History, 27.11.2020 22:50

Mathematics, 27.11.2020 22:50


Mathematics, 27.11.2020 22:50





Mathematics, 27.11.2020 22:50

Mathematics, 27.11.2020 22:50


Mathematics, 27.11.2020 22:50

English, 27.11.2020 22:50

Arts, 27.11.2020 23:00


Biology, 27.11.2020 23:00




