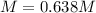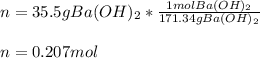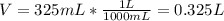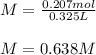Chemistry, 27.01.2021 08:00 quanntwann2873
What is the molarity of a solution that is made by mixing 35.5 g of Ba(OH)2 in 325 ml of solution?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:50
What are four significant sources of ghgs that come from wostem washington?
Answers: 2

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 14:30
What state of matter is ice a. liquid b. element c. solid d. gas
Answers: 1

Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1
You know the right answer?
What is the molarity of a solution that is made by mixing 35.5 g of Ba(OH)2 in 325 ml of solution?...
Questions


Mathematics, 29.03.2021 19:40





Advanced Placement (AP), 29.03.2021 19:40

Chemistry, 29.03.2021 19:40

Mathematics, 29.03.2021 19:40

Mathematics, 29.03.2021 19:40

Mathematics, 29.03.2021 19:40

English, 29.03.2021 19:40

Mathematics, 29.03.2021 19:40


Mathematics, 29.03.2021 19:40

Mathematics, 29.03.2021 19:40

Business, 29.03.2021 19:40