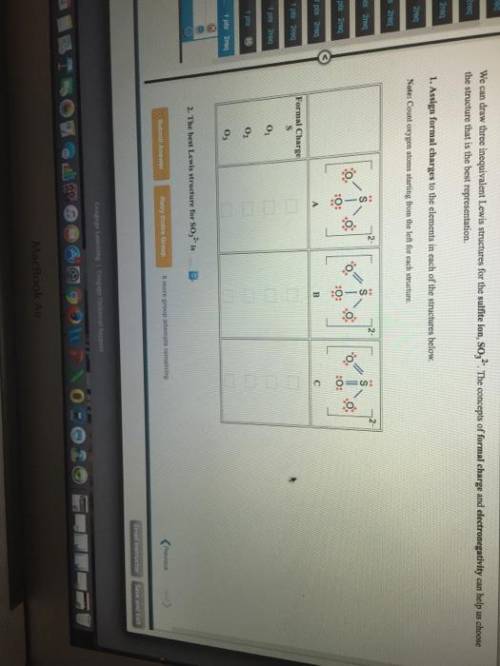
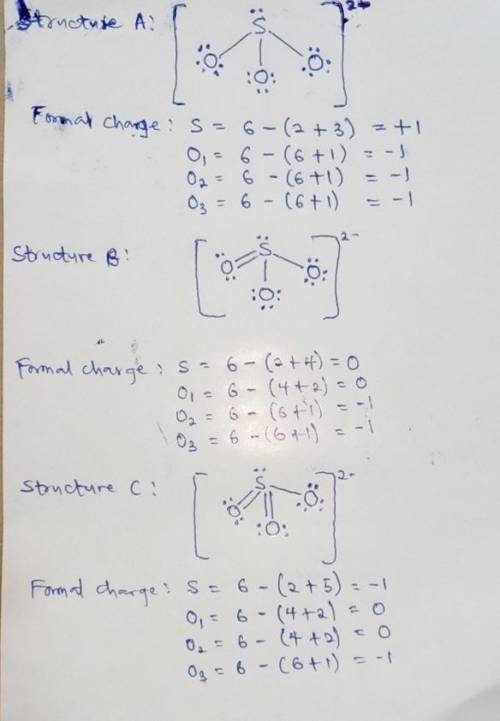
We can draw three inequivalent Lewis structures for the sulfite ion , SO32- . The concepts of formal charge and electronegativity can help us choose the structure that is the best representation. 1. Assign formal charges to the elements in each of the structures below. Note: Count oxygen atoms starting from the left for each structure. A B C Formal Charge S O1 O2 O3 2. The best Lewis structure for SO32- is

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2


Chemistry, 22.06.2019 13:00
Asubstance is a good conductor of electricity which of the following best explains a probable position of the substance in a periodic table
Answers: 3

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3
You know the right answer?
We can draw three inequivalent Lewis structures for the sulfite ion , SO32- . The concepts of formal...
Questions


Mathematics, 07.04.2020 21:45

Physics, 07.04.2020 21:45


Mathematics, 07.04.2020 21:45



Advanced Placement (AP), 07.04.2020 21:45

English, 07.04.2020 21:45

History, 07.04.2020 21:45




Biology, 07.04.2020 21:45

Biology, 07.04.2020 21:45

Mathematics, 07.04.2020 21:45



History, 07.04.2020 21:45

Mathematics, 07.04.2020 21:45