
Chemistry, 28.01.2021 22:10 victorialeverp714lg
The chemical equation for the process of photosynthesis is shown below. Which part represents the chemical energy that is formed in the process?
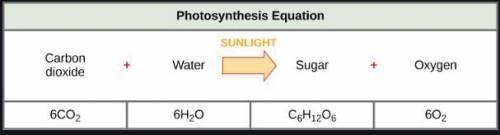

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:40
How many liters of hydrogen gas will be produced at stp from the reaction of 7.179×10^23 atoms of magnesium with 54.219g of phosphoric acid (h3po4) the equation is 3mg + 2h3(> mg(po4)2+3h2
Answers: 1

Chemistry, 22.06.2019 05:30
Arecipe calls for 1.2 cups of oil. how many liters of oil is this?
Answers: 2

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

You know the right answer?
The chemical equation for the process of photosynthesis is shown below. Which part represents the ch...
Questions


Computers and Technology, 08.03.2021 22:30

Business, 08.03.2021 22:30

Mathematics, 08.03.2021 22:30

Social Studies, 08.03.2021 22:30

Mathematics, 08.03.2021 22:30

Mathematics, 08.03.2021 22:30


English, 08.03.2021 22:30

Mathematics, 08.03.2021 22:30




Mathematics, 08.03.2021 22:30

Mathematics, 08.03.2021 22:30


Computers and Technology, 08.03.2021 22:30

Arts, 08.03.2021 22:30




