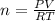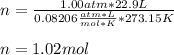Chemistry, 29.01.2021 14:00 aisatubrodie4626
50POINTS!
Using the ideal gas law (PV=nRT) solve for the missing. Variable. R= 0.08206atm*L/mol*k
If 22.9L of an ideal gas was collected at STP. How many moles of the gas were present?
A. 1.02 moles
B. 5.99 moles
C. 3.05 moles
D. 2.74 moles


Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3
You know the right answer?
50POINTS!
Using the ideal gas law (PV=nRT) solve for the missing. Variable. R= 0.08206atm*L/mol*k
Questions



Mathematics, 30.06.2021 04:30








Chemistry, 30.06.2021 04:30

Mathematics, 30.06.2021 04:30





Computers and Technology, 30.06.2021 04:30

Mathematics, 30.06.2021 04:30

Mathematics, 30.06.2021 04:30

Mathematics, 30.06.2021 04:30