Chemistry, 29.01.2021 16:50 mcaninch36
Consider the combustion of propane: A balloon is being inflated to its full extent by heating the air inside it. In the final stages of this process, the volume of the balloon changes from L to L by the addition of J energy as heat. Assume that all the heat comes from the combustion of propane. What mass of propane must be burned to furnish this amount of energy assuming the heat transfer process is 50.% efficient?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no2]2 ? second order 3/2 order third order zero order none of the listed answers are correct
Answers: 3

Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2

Chemistry, 22.06.2019 19:50
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3

Chemistry, 22.06.2019 22:30
What methods could you use to solubilize calcium carbonate
Answers: 1
You know the right answer?
Consider the combustion of propane: A balloon is being inflated to its full extent by heating the ai...
Questions



Mathematics, 24.05.2020 05:00



Mathematics, 24.05.2020 05:00


Mathematics, 24.05.2020 05:57

Social Studies, 24.05.2020 05:57


Mathematics, 24.05.2020 05:57

Spanish, 24.05.2020 05:57



Chemistry, 24.05.2020 05:57


Mathematics, 24.05.2020 05:57

Biology, 24.05.2020 05:57


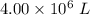 to
to 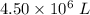 .
.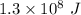
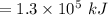
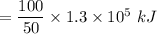
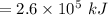
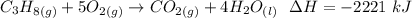
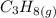
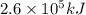 will produce
will produce