
Chemistry, 01.02.2021 14:00 NerdyJason
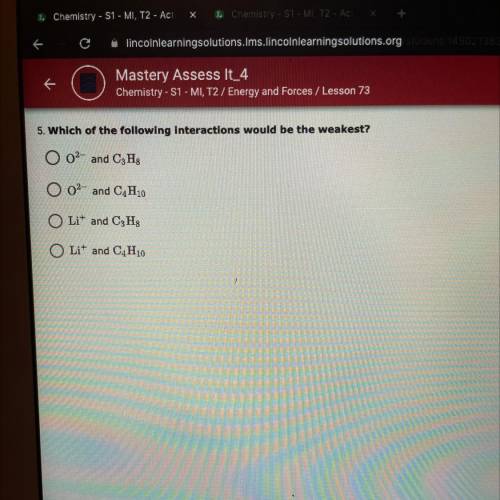
Which of the following interactions would be the weakest? Picture provided, 10 points. Please only answer correctly.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:00
What is the subscript for oxygen in its molecular formula
Answers: 1

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
Which of the following interactions would be the weakest?
Picture provided, 10 points. Please only...
Questions

Mathematics, 19.07.2019 09:00

Mathematics, 19.07.2019 09:00

Mathematics, 19.07.2019 09:00

Mathematics, 19.07.2019 09:00

Mathematics, 19.07.2019 09:00


Mathematics, 19.07.2019 09:00

Mathematics, 19.07.2019 09:00


Biology, 19.07.2019 09:00

Biology, 19.07.2019 09:00

Mathematics, 19.07.2019 09:00

History, 19.07.2019 09:00

Social Studies, 19.07.2019 09:00

History, 19.07.2019 09:00

Biology, 19.07.2019 09:00

Chemistry, 19.07.2019 09:00

History, 19.07.2019 09:00

History, 19.07.2019 09:00

Chemistry, 19.07.2019 09:00



